|
Once neutralised, the particles are no longer. 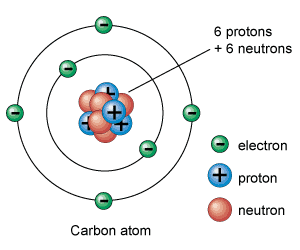
The only difference between an atom and its ions is the number of electrons that surround the nucleus. The gain or loss of electrons by an atom to form negative or positive ions has an enormous impact on the chemical and physical properties of the atom. In fact, the negative ion can be more than twice as large as the neutral atom. By adding one more electron we get a negatively charged Cl-ion with a net charge of -1. Having eight 3d electrons and two 4s electrons is much less energetically stable than ten 3d electrons and no 4s electrons.\): The three most stable isotopes of hydrogen. Neutral atoms resulting from the capture of free electrons by positively ionized nuclei of the solar wind. A neutral chlorine atom, for example, contains 17 protons and 17 electrons. Atoms are therefore neutral in their normal states. When d-block elements lose electrons, they lose the highest energy s electrons first, which in the case of zinc are the two 4s electrons. When the number of protons equals the number of electrons, we call the atom a neutral atom. For the d-block elements, the outermost s-sublevel has higher energy than the d-sublevel, which is contrary to what the Aufbau diagram indicates. Zinc is a d-block element, also known as a transition element. Our experiment features a multi-section chamber, atoms are first in a 3D MOT in the octagon chamber. And this is explained if we look at fluorine's valence shell (the second shell) electron configuration: 2s (2)2p (5). For instance, if we take an atom of fluorine, F, in it's neutral state it has no net charge however it is VERY reactive. The #"Zn"^(2 )# ion has lost two electrons, which leaves it with 30 protons and 28 electrons. So even if an atom is neutral, that is not necessarily its most stable state. We can calculate an atom's formal charge using the equation FC VE - LPE - (BE), where VE the number of valence electrons on the free atom, LPE the number of lone pair electrons on the atom in the molecule, and BE the number of bonding (shared) electrons around the atom in the molecule. Grey dashed lines of various styles indicate different neutral atom populations, such as the ISN flow (ANAs), Ribbon ENAs, secondary IS neutral atoms. The mass number of an atom is calculated by adding together the number of protons and neutrons that are found within that atom, as shown below. Therefore, every atom of tungsten also contains 74 electrons. After the formation of the electrically conducting carbonnitrogen. The formation of the N/C framework with coordinating nitrogen sites is performed using zinc as a templating agent. 
Atoms that gain extra electrons become negatively charged. By removing an electron from this atom we get a positively charged Na ion that has a net charge of 1. A neutral sodium atom, for example, contains 11 protons and 11 electrons. The electron configuration of a neutral zinc atom is #"1s"^2"2s"^2"2p"^6"3s"^2"3p"^6"3d"^10"4s"^2"#. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. We examine the performance of a number of single-atom MN/C electrocatalysts with a common structure in order to deconvolute the activity of the framework N/C support from the metal MN4 sites in MN/Cs. Neutral atoms can be turned into positively charged ions by removing one or more electrons. Atoms with lesser electrons than protons are positively charged and. Atoms with more electrons than protons are negatively charged and are known as anions. They have an unequal number of protons and electrons. 
A neutral atom has equal numbers of protons and electrons, so a neutral atom of zinc would have 30 electrons. Neutral Atom vs Charged Atom Charged atoms are known as ions. Since it is the electrons that determine how one atom interacts with another, in the end it is the number of protons in the nucleus that determines the chemical properties of an atom. Arrays of isolated neutral atoms show promise for quantum computing due to neutral-atom qubits being well isolated from environmental noise and being highly. The atomic number of zinc is 30, which means that all zinc atoms have 30 protons in their nuclei. A neutral atom has an equal number of protons and electrons so that the positive and negative charges exactly balance.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
